SEMAX
Definition
Semax is a peptide drug based on a fragment of the adrenocorticotropic hormone (ACTH). In Russia, Semax is used as a medication for the prevention and treatment of circulatory disorders including stroke. Semax is listed on the Russian List of Vital & Essential Drugs, a reference of key medications which is reviewed and approved by the Russian government. Semax is also used as a nootropic, or cognitive enhancer, due to evidence that Semax can enhance memory performance and reduce the negative effects of stress.
Semax’s action mechanisms are not entirely clear, but research has suggested that much of its neuroprotective and restorative effects may be traced to the activation of the brain serotoninergic system in addition to increased BDNF expression in the hippocampus.[4] In addition, the Semax peptide is known to affect several biological processes involved in the function of various systems. The peptide markedly affects the immune response, altering the expression of genes that modulate the amount and mobility of immune cells and enhancing the expression of genes that encode chemokines and immunoglobulins.[12] In studies conducted on rats, Semax is also observed to influence the expression of genes that promote the formation and functioning of the vascular system.[12] As a result, it is believed that the key mechanisms of the peptide’s neuroprotective effects are likely to be its immunomodulating effect and its impact on the vascular system during ischemia (inadequate blood flow).[12] Moreover, a 2006 study published in Brain Research concluded that the Semax peptide affects cognitive brain functions by modulating the expression and the activation of the hippocampal BDNF/trkB system.[5] Researchers in the study analyzed the effects of Semax administration on the brains of rat subjects, observing that a 1.4-fold increase of BDNF protein levels along with a 1.6-fold increase of trkB tyrosine phosporylation levels occurred.[5] Additionally, a 3-fold increase of exon III BDNF and a 2-fold increase of trkB mRNA levels occurred in the rat hippocampus as well. This supports the view that activation of the hippocampal BDNF/trkB system is key to the action mechanisms of Semax.[5] A recent 2017 study published in Molecular Genetics and Genomics demonstrated Semax’s powerful protective effect in rat subjects with ischemic brain injury (brain stroke).[9] Researchers noted that the peptide markedly affected the rats’ immune response.[9] The Semax peptide was observed to enhance the antigen presentation signaling pathway, intensify ischemia’s effect on the interferon signaling pathways, and affect the processes for synthesizing immunoglobulins.[9] Additionally, the peptide was seen to significantly increase expression of the gene encoding the immunoglobulin heavy chain.[9] Study researchers postulated that Semax’s neuroprotective mechanism was achieved through “neuroimmune crosstalk” and newly identified properties of Pro-Gly-Pro (PGP), a component of the Semax peptide.[9] Moreover, further research has indicated that decreased vascular injury and enhanced mRNA neurotrophin synthesis occurs after Semax administration in rats that have suffered cerebral ischemia due to stroke.[10] In a 2013 study, Semax was observed to have anti-inflammatory properties which manifested in reduced levels of vascular endothelial growth factor (VEGFA) in rat study subjects after ischemic brain injury.[10] In 2007, researchers published a paper in Medical Hypothesis suggesting Semax as a potential future treatment for Attention-Deficit Hyperactivity Disorder (ADHD) and Rett Syndrome.[2] Researchers noted that evidence gleaned from previous animal studies suggested that Semax was able to augment the effects of psychostimulants on central dopamine release while additionally stimulating central brain-derived neurotrophic factor (BDNF) synthesis.[2] Importantly, it was also noted in previous study that Semax could improve selective attention and modulate brain development.[2] As a result, they suggested that the increased central BDNF activity as a result of Semax administration could prove highly therapeutic in regards to Rett syndrome, while noting that BDNF and increased dopamine release could also highly benefit cases of ADHD. Remarkably, Semax has exhibited cognitive effects in animal studies that seem to go far beyond protection and recovery from acute brain injury. In fact, studies have shown it to have a measureable effect on animal behavior. A 2007 study analyzed the effects of the Semax peptide on the exploratory activity, anxiety level, and depression-like behavior in white rats.[4] While it was shown that chronic Semax administration did not significantly influence exploratory activity of the rats in a “non-stressogenic environment,” it did however stimulate anxiolytic (anti-anxiety) and antidepressant effects.[4] Researchers in the study concluded that the results gained from Semax administration may be traced to the activation of the brain serotoninergic system as well as to increased BDNF expression in the hippocampus.[4] In addition, the Semax peptide has been shown to reduce memory and learning deficits in rat study subjects treated with amphetamines in utero.[11] In this study, published in BMC Pharmacology, it was observed that Semax induced delayed memory deficits and significant learning impairments in juvenile offspring of rats.[11] Therefore, researchers concluded that the peptide may lead to a significant recovery of the memory functions of brain-damaged rat subjects, opening new approaches for neuroprotection and cognitive rehabilitation of prenatal brain damage.[11] Further, a 2001 study examined the effects of Semax administration on subjects with glaucomatous optic neuropathy, an eye disorder.[6] Intriguingly, it was shown by electrophysiological and computer methods of examination that the Semax peptide outperformed traditional neuroprotective treatments for glaucoma. Researchers concluded that the success of Semax was due to its powerful neuroprotective and neurotrophic effects, further demonstrating the potent healing effect the peptide seems to exhibit on the neurological system.[6] The neuroprotective properties of Semax peptides have been demonstrated in animal studies to protect against damaging effects of heavy metal poisoning as well. A recent 2016 study published in Doklady Biological Sciences observed the effects of Semax on heavy metal poisoning in rats compared with ascorbic acid.[8] Researchers established that heavy metal salts inhibited the avoidance response in rat subjects, and that Semax counteracted this effect as strongly as ascorbic acid.[8] As a result, researchers confirmed the peptide’s antioxidant properties.[8] Moreover, the Semax peptide has been shown to moderate the effects of copper cytotoxicity in animal studies as well.[7] Researchers in a 2015 study published in the Journal of Inorganic Biochemistry noted that Semax exhibits marked neuroprotective activities on cognitive brain functions.[7] Dys-homeostasis of metal ions is involved in several neurodegenerative disorders, so it was hypothesized that Semax would be effective in mitigating these disorders.[7] Ultimately, they found that a reduced copper induced cytotoxicity was indeed observed following Semax administration.[7] Certainly, clinical study has shown that Semax’s possible therapeutic value may well extend beyond mental and cognitive disorders. In a 2002 study published in the Bulletin of Experimental Biology and Medicine, researchers studied the effects of the Semax peptide on healing peptic ulcers.[3] Remarkably, it was observed that ulcer healing occurred in 89.5% patients receiving intranasal Semax compared to just over 30% in the control group.[3] Researchers did note that further study of Semax in different combinations with common antiulcer drugs was needed, but these findings proved quite promising for future therapeutic applications.[3]
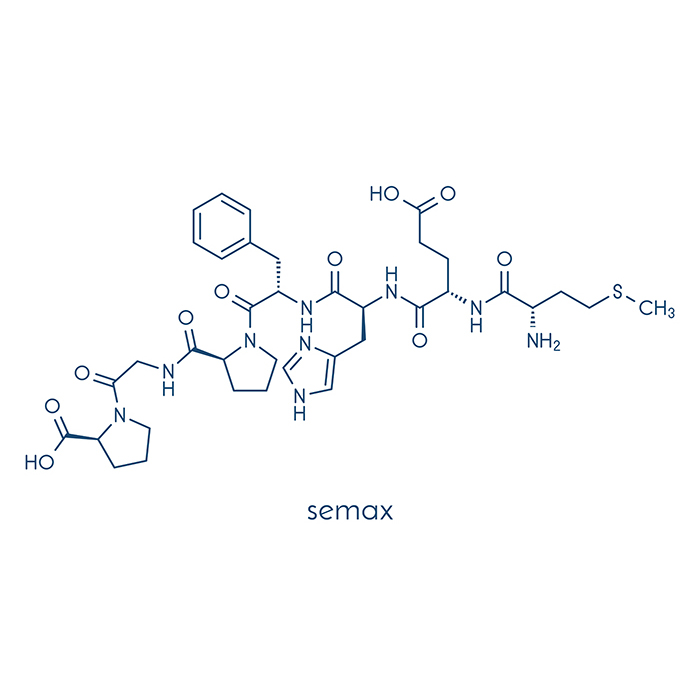
Specs
Molar mass: 813.92 g/mol
Molecular formula: C37H51N9O10S
Sequence: Met-Glu-His-Phe-Pro-Gly-Pro
CAS: 4037-01-8
Positive effects
1) May Prevent Oxidative Damage
Semax has been shown to protect the body and brain from various types of oxidative damage/stress. In animal studies, Semax has been shown to prevent oxidative damage caused by the poisonous effects of lead exposure in the brain. Semax has been shown to prevent the oxidative liver damage which occurs with chronic stress in lab animals. Semax also prevents oxidative damage to the body after a stroke or heart attack. Researchers have suggested that this improvement is in part a result of oxidative protection.
2) May Improve Memory
Semax improved memory and attention in a Russian study of task performance in healthy men. Semax also decreased learning time in animal studies. Some researchers have suggested that the resistance to oxidative damage in the brain and improved brain circulation might explain the improved memory seen in these subjects.
3) May Improve Circulation
Semax can be used in the treatment of ischemic stroke in patients. In rats, it is capable of protecting the heart from damage after a stroke or heart attack. The drug partially prevents the growth of the heart caused by high blood pressure, thus potentially preventing heart failure development after a heart attack. A major risk factor for stroke is chronically poor circulation in the brain, a condition medically known as cerebrovascular insufficiency. Patients with different stages of cerebrovascular insufficiency saw significant stabilization of disease progression and reduced risk of stroke when treated with Semax in one study (retrospective review). Researchers attributed the results in part to improved circulation.
4) May Lower the Negative Effects of Stress
Semax has been shown to decrease the negative physical and mental responses to stress in animal studies. One negative physical response to chronic stress is the increased production of specific liver enzymes. Treatment with Semax was shown to prevent their increase in several animal studies. Semax normalized behavioral changes associated with increased stress resulting from physical injury. Semax also normalized behavioral changes associated with increased stress due to developmental problems in animals.
5) Increases BDNF
Semax increases the brain levels of a substance known as the brain-derived neurotrophic factor (BDNF). BDNF is a major chemical factor that is essential for cognitive development, synaptic plasticity, and neuronal survival. Studies indicate that low levels of BDNF may also cause or worsen depression and anxiety. By increasing the level of BDNF, Semax could help alleviate anxiety and depression. Animal studies have also indicated that Semax normalizes behavior associated with increased stress and anxiety, possibly as a result of this mechanism. In a study (randomized controlled trial) of patients recovering from the stroke, Semax improved restoration of nerve function in subjects relative to those that did not receive the same treatment. This improvement in nerve function may be a result of increased BDNF.
6) May Help Alleviate Pain
Semax prevents the breakdown of chemicals called enkephalins which regulate the response to pain. An increase in enkephalins may reduce the severity of perceived pain.
7) May Help ADHD Symptoms
Semax has a number of effects that may be helpful in the treatment of ADHD, including memory improvement and the increase of neurotransmitters which are often lower in patients with this condition. Researchers have proposed evaluating Semax as a treatment for ADHD, and anecdotes support that Semax is occasionally used in Russia for this purpose. Additional studies are needed to determine if there is a benefit of Semax in the treatment of ADHD. Also, proper dosage and treatment strategy would need to be determined.
Advanced science
- Increases brain BDNF levels.
- Prevents the breakdown of enkephalins.
- Protects the nervous system from oxidative damage.
- Affects the expression of genes related to the immune and vascular systems during stroke.
Side effects
Semax is not FDA approved and has not been evaluated by the FDA. It has been featured in numerous clinical trials on humans and all of these tests have reported no side effects or hormonal activity . One study in animals, using a high concentration of Semax, found that it had an anti-oxidant effect at low dosages, but a pro-oxidant effect at higher dosages. Another animal study using various dosages found that each dosage studied had antioxidant effects. More research needs to be done to determine if there is a negative effect associated with high dosages, and at what dosage that effect occurs if present. Some users have reported increased anxiety as a side effect of using Semax as a nootropic.
Purely for Scientific Research
Despite the fact that there has been plenty of research on Semax in relation to how it functions and the benefits that can be derived from such functionality, it should be emphasized that the peptide is still just intended for scientific study at this point in time. Because of this, any findings or observations relating to Semax overall functionality, mechanics, benefits, should exclusively be the product of study performed in a strictly contained environment.
References
1. Biophysical Journal. Volume 95. November 2008 4879–4889. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2576403/pdf/4879.pdf
2. Med Hypotheses. 2007;68(5):1144-6. Epub 2006 Sep 25. https://www.ncbi.nlm.nih.gov/pubmed/16996699
3. Ivanikov, I.O., Brekhova, M.E., Samonina, G.E. et al. Bulletin of Experimental Biology and Medicine (2002) 134: 73. doi:10.1023/A:1020621124776. https://link.springer.com/article/10.1023%2FA%3A1020621124776
4. Ross Fiziol Zh Im I M Sechenova. 2007 Jun;93(6):661-9. https://www.ncbi.nlm.nih.gov/pubmed/17850024
5. Brain Res. 2006 Oct 30;1117(1):54-60. Epub 2006 Sep 22. https://www.ncbi.nlm.nih.gov/pubmed/16996037
6. Vestn Oftalmol. 2001 Jul-Aug;117(4):5-8. https://www.ncbi.nlm.nih.gov/pubmed/11569188
7. J Inorg Biochem. 2015 Jan;142:39-46. doi: 10.1016/j.jinorgbio.2014.09.008. Epub 2014 Sep 28. https://www.ncbi.nlm.nih.gov/pubmed/25310602
8. Dokl Biol Sci. 2016 May;468(1):112-4. doi: 10.1134/S0012496616030066. Epub 2016 Jul 14. https://www.ncbi.nlm.nih.gov/pubmed/27411820
9. Mol Genet Genomics. 2017 Jun;292(3):635-653. doi: 10.1007/s00438-017-1297-1. Epub 2017 Mar 2. https://www.ncbi.nlm.nih.gov/pubmed/28255762
10. Mol Biol (Mosk). 2013 May-Jun;47(3):461-6. https://www.ncbi.nlm.nih.gov/pubmed/23888777
11. BMC Pharmacol. 2009; 9(Suppl 2): A26. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2778896/
12. BMC Genomics. 2014; 15: 228. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3987924/








